Featured Scientist: Pascal Nnaemeka Eyimegwu, M.S. 2019, Department of Chemistry , Illinois State University

Birthplace: Nkalagu, Nigeria
My Research: I study how to make reactions go faster using nanoparticles.
Research Goals: I would like to continue to study nanoparticles (tiny materials) and how they might be used to speed up chemical reactions. This research would help scientists make medicine more efficiently.
Career Goals: I want to be a Professor of Chemistry.
Hobbies: I like to travel and play soccer.
Favorite Thing About Science: Science is interesting because we can use it to interpret everything around us. It provides solutions to our problems. It’s no wonder that we say “all in nature is chemistry”.
Organism of Study: Gold nanoparticles

Field of Study: Analytical Chemistry
What is Analytical Chemistry? Analytical Chemistry focuses on identifying and quantifying chemicals. For example, analytical chemists are responsible for making sure that your food and water are safe to eat and drink. Analytical Chemistry has been used to develop tools that can check if drivers are under the influence of alcohol, or to tell the difference between illegal drugs and medicines. In addition, this form of chemistry is used to check the quality of food and drugs to make sure that they are safe.
Check Out My Original Paper: “Atypical catalytic function of embedded gold nanoparticles by controlling structural features of polymer particle in alcohol-rich solvents”

Citation: P. N. Eyimegwu, J.H. Kim, Atypical catalytic function of embedded gold nanoparticles by controlling structural features of polymer particle in alcohol-rich solvents. J. Nanotechnol. 30.28 (2019).
Research at a Glance: The goal of this study was to make stable catalysts that can speed up the chemical reactions used to make starting materials in pharmaceutical industries. A catalyst is a substance that makes a reaction go faster. The type of catalyst that I work with can be recycled multiple times without losing its efficiency, unlike the most common type, which can only be used once. In this research, gold nanoparticles are used as the catalysts. However, these nanoparticles attract each other and need to be stabilized to be used effectively. In my study, I stabilize gold nanoparticles using a specialized polymer, an organic compound used in making plastics. The catalyst that I made was highly stable, reactive, and produced no undesired by-products. The catalysts that I designed in my study can be used to produce important starting materials to organic compounds that are often used in pharmaceutical industries.
Highlights: The catalyst (gold nanoparticles in a polymer) that I made can be used to produce biphenyl, which is a useful starting material for making many organic compounds. I tried to use many different solvents to make biphenyl. The turning point of my research was in the decision to change the solvent for my catalytic reaction. The first solvent that I tried to use was water, but it resulted in poor yields of my target product, biphenyl. Ethanol was able to solve this problem.
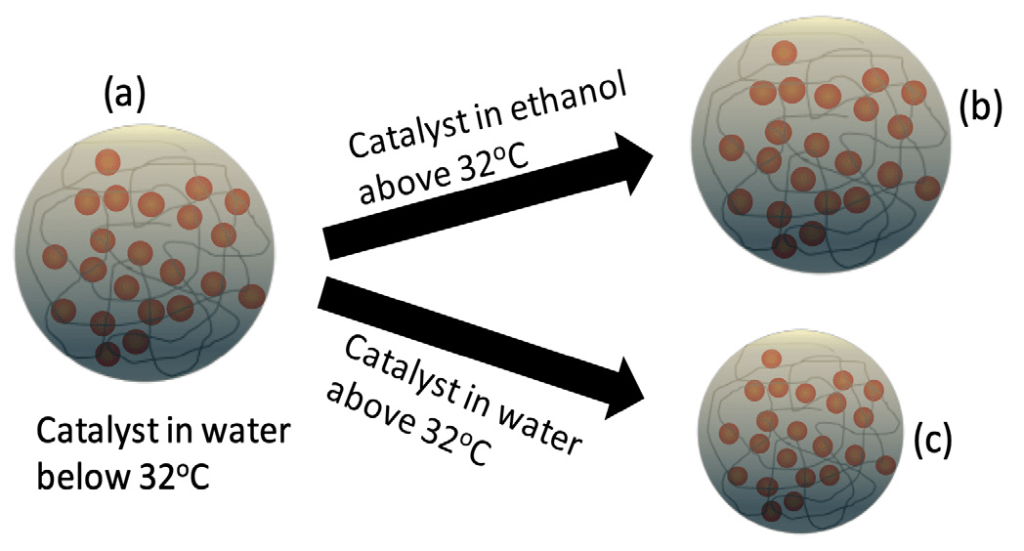
The gold nanoparticles are used as the catalyst. The image on the left (a) shows the structure of the catalyst in water below 32°C. The images on the right show the catalyst in ethanol (b) and in water (c) above 32°C. As you can see, the catalyst in water looks smaller but remains swollen and slightly bigger in ethanol.
Why was ethanol the trick? Unlike water, ethanol tends to remove the organic compounds that prevent the catalyst from performing well. Additionally, I used a polymer in my reaction. The polymer swells more when it is in ethanol than when it is in water, which guarantees more movement of reactants and products in and out of the polymer. The reaction with water creates a by-product, but when I used the ethanol, the by-product was no longer produced. Finally, I found out that the yield is highest when I used ethanol (Figure 1). This is because ethanol can remove things from the catalyst that hinder the reaction.
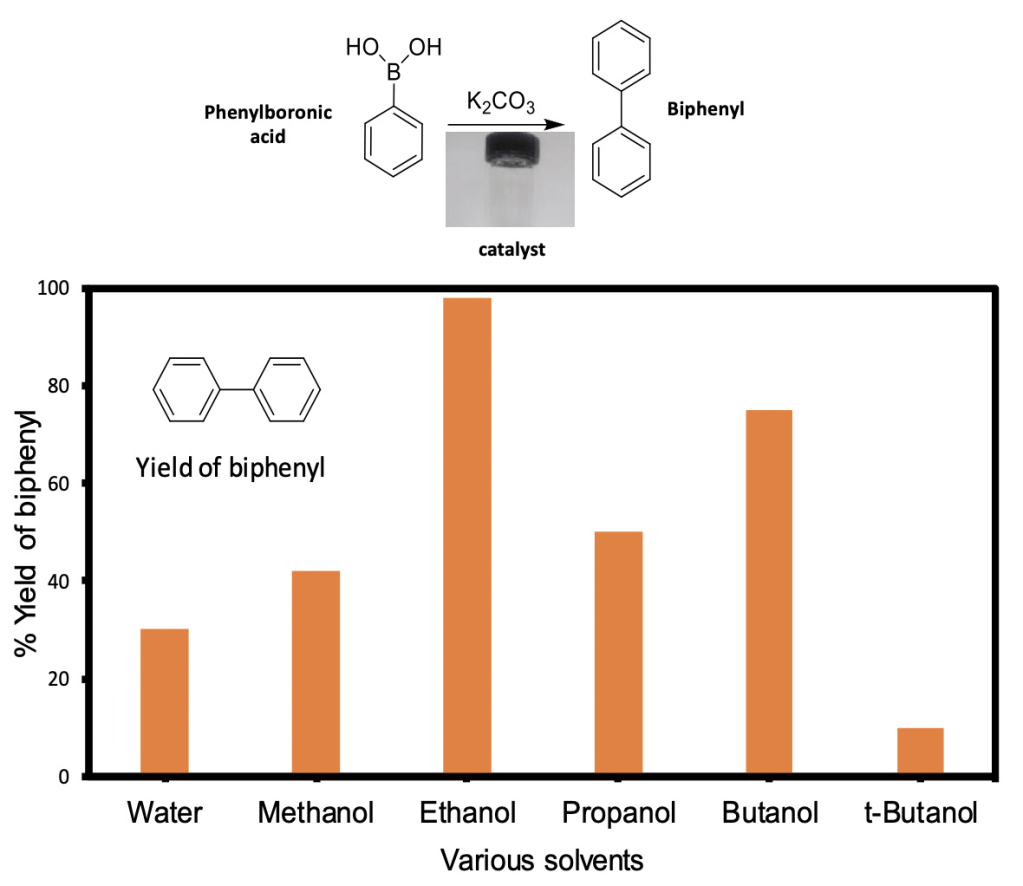
Figure 1. Production of biphenyl using phenylboronic acid as the starting material. The x-axis shows the solvents that I used in the catalytic reaction.The y-axis shows how much biphenyl was made. The orange bars represent the amount of biphenyl that was produced after a reaction in each individual solvent. Adapted from Eyimegwu et al. 2019.
Next, I wanted to make sure that my catalyst was stable. To do this, I needed to run a test to make sure that my catalyst was recyclable. Being recyclable means that the same catalyst can be used to speed up different batches of reactions without losing its strength. The first, second, and third cycle gave the same yield, but the yield went down on the fourth cycle and went down further on the fifth cycle. I was able to bring it up again after purifying it in water and putting it back into ethanol for the 6th cycle (Figure 2).
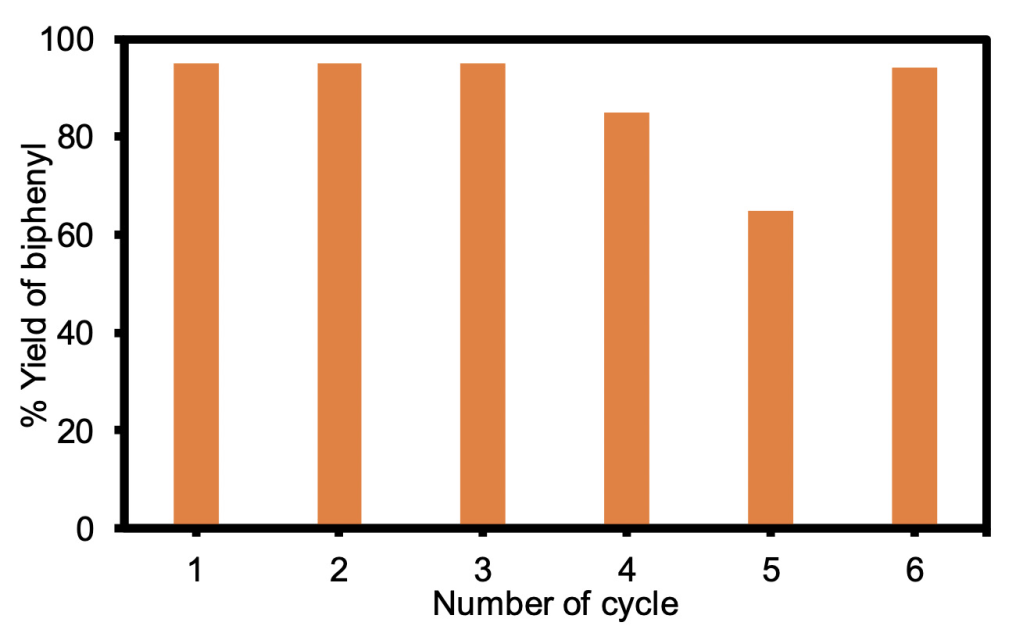
As you can see in Figure 2, the catalyst produced just as much biphenyl after the 6th reaction as it did in the 1st reaction. This means that my catalyst should not be thrown away after one reaction because it will still be useful in another one.
What My Science Looks Like: The image below has two solutions. The one on the left (A) shows gold nanoparticles stabilized by a polymer, so that they don’t clump together. The one on the right (B) shows the gold nanoparticles by themselves. These are the types of solutions that I work with in my research.

The next image shows the steps that I used to carry out the chemical reaction for my experiment. The goal was to make my target product, biphenyl.
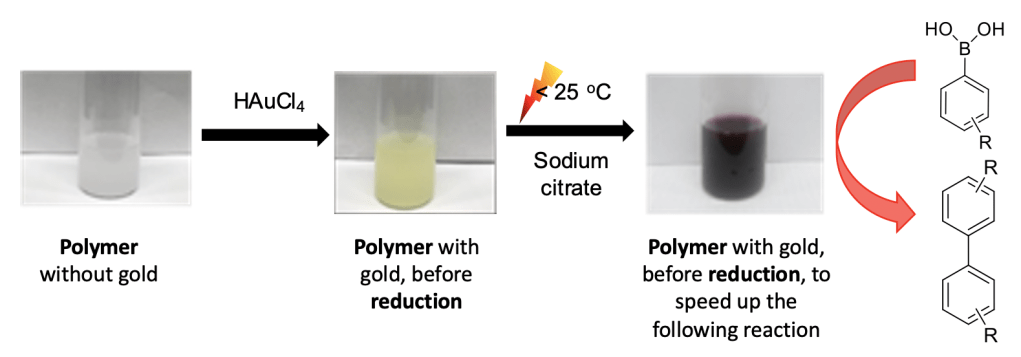
The Big Picture: Gold nanoparticles are safe and inexpensive. People are working hard to use them to make chemical reactions happen more quickly. My research introduces interesting properties of these nanoparticles: they change their catalytic activity when they are put in a polymer and are introduced to ethanol. The products of these reactions can be used to make medicines.
Decoding the Language:
Biphenyl: A useful starting material for making many organic compounds used by pharmaceutical companies.
Catalyst: Materials that make reactions go faster
Gold solution: The first state of gold, before it is reduced to form particles.
Nanoparticles: Particles between the range of 1-100 nm in size.
Organic compounds: A chemical compound is made up of two or more elements. For example, table salt (NaCl) is a chemical compound because it is made up of both sodium (Na) and chlorine (Cl). An organic compound is any chemical compound that contains carbon. For example, sugar (C6H12O6) is an organic compound. It is made up of carbon (C), hydrogen (H), and oxygen (O).
Phenylboronic acid: A chemical and the starting material that I used to make biphenyl.
Polymer: An organic compound used in making plastics and nylon. In my research, I used it to stabilize gold nanoparticles so that the gold nanoparticles would not clump together.
Reactants: The materials that come together to produce another material.
Recyclable catalyst: A catalyst that can be used more than once.
Reduction: A way of making gold solution to become particle.
Solvent: A liquid that is used to carry out a reaction or to dissolve a solid material. Water is an example of a solvent.
Products: The result of a reaction, what we are trying to make.
Learn More:
Another research paper on catalysis: P.N. Eyimegwu, J.A. Lartey, J.H. Kim, GoldNanoparticle-Embedded Poly (N-isopropylacrylamide) Microparticles for Selective Quasi-Homogeneous Catalytic Homocoupling Reactions. ACS Appl. Nano Mater. 2.9: 6057-6066 (2019) .
Synopsis written by: Rosario Marroquin-Flores, PhD (Anticipated Spring 2022), School of Biological Sciences, Illinois State University
Download this article here
Please take a survey to share your thoughts about the article!
